In a decision clarifying the boundaries between claim construction and factual findings of inherency, the Federal Circuit in Sigray, Inc. v. Carl Zeiss X-Ray Microscopy, Inc., No. 23-2211 (Fed. Cir. May 23, 2025), reversed the PTAB’s determination that certain claims of U.S. Patent No. 7,400,704 were not anticipated by the prior art. The court concluded that the Board had improperly narrowed the scope of the claims through implicit construction and that, under the correct claim scope, the prior art reference Jorgensen inherently disclosed the disputed limitation.
Background
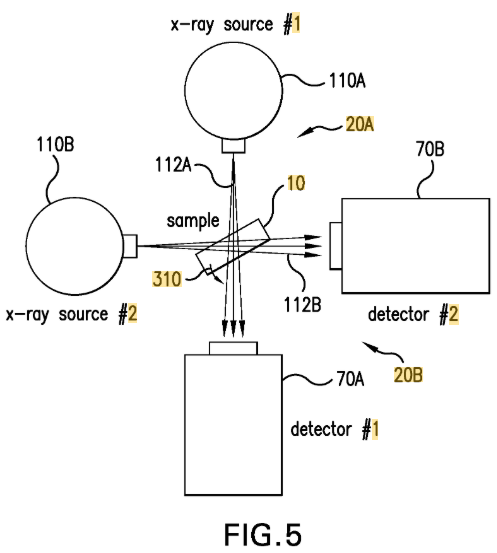
Zeiss’s patent claims an X-ray imaging system incorporating “projection magnification,” with the key limitation being that the magnification of the projection stage is “between 1 and 10 times.” Sigray petitioned for inter partes review, arguing that a 1998 paper by Jorgensen disclosed all limitations of the claims, including this magnification range.
The Board acknowledged that Jorgensen disclosed nearly all elements of the claims but found no anticipation because it concluded that the reference did not disclose “enough” beam divergence to result in the required projection magnification. Sigray appealed, arguing that this conclusion was based on an implicit and erroneous narrowing of the claim scope.
Implicit Claim Construction
The Federal Circuit found that the Board had implicitly construed the phrase “between 1 and 10 times” in a way that excluded very small—indeed undetectable—levels of magnification. The Board’s repeated focus on whether Jorgensen’s beam diverged “enough” and whether it created a “meaningful” amount of magnification revealed that it was assessing not just whether any magnification was present, but whether the magnification was perceptible or functionally significant.
As the court explained, “[t]he Board’s use of the word ‘enough’ reflects that it considered a certain level of divergence as outside the claim. Narrowing the claim scope in this way is in fact claim construction.” The court emphasized that claim construction had occurred even though the Board disclaimed doing so—relying on its precedent in Google LLC v. EcoFactor, Inc., 92 F.4th 1049 (Fed. Cir. 2024), to look at the Board’s analysis and outcome rather than its labels.
Inherent Disclosure and Physical Geometry
After correcting the Board’s construction, the court held that Jorgensen inherently disclosed the disputed magnification limitation. Charles Gideon Korrell sees that the opinion stresses that under the geometric optics formula (M = (Ls + Ld)/Ls), any system with a diverging X-ray beam and a nonzero sample-to-detector distance (Ld > 0) necessarily results in magnification greater than 1.
Since perfect collimation—i.e., zero divergence—is physically impossible in real-world systems, the court found that Jorgensen’s setup, which necessarily included some divergence, inherently satisfied the “between 1 and 10 times” magnification requirement. As stated in the opinion: “Here, it is undisputed that Jorgenson’s X-ray beams are not completely parallel and naturally must result in some magnification. That miniscule amount of magnification disclosed by the prior art definitionally achieves a magnification within the claimed range of 1 to 10.”
Charles Gideon Korrell notes that the court relied heavily on SmithKline Beecham Corp. v. Apotex Corp., 403 F.3d 1331 (Fed. Cir. 2005), in concluding that inherent anticipation does not require recognition or intention by the prior art. Rather, it is sufficient that the claimed feature necessarily results from practicing the prior art reference, regardless of whether it was appreciated at the time.
Reversal and Remand
- Claims 1, 3, and 4: Reversed. The court found that these claims were inherently anticipated by Jorgensen.
- Claims 2, 5, and 6: Vacated and remanded. Sigray had argued these claims were obvious, not anticipated, so the Board must evaluate obviousness in light of the Federal Circuit’s clarified claim scope.
Takeaway
Charles Gideon Korrell thinks that this decision illustrates the Federal Circuit’s firm stance on the plain meaning of claim terms. The phrase “between 1 and 10 times” includes any magnification over 1, no matter how small, and the Board erred by requiring a “meaningful” or “detectable” amount. The ruling reinforces the principle from SmithKline that inherent disclosure encompasses all inevitable consequences of prior art, even if imperceptible.
It also underscores how implicit claim construction—especially when it narrows the scope based on technical judgments about magnitude or significance—can fundamentally alter the outcome of IPR proceedings. Courts and the PTAB alike must be careful not to impose unstated thresholds that conflict with the express language of the claims.
Finally, Charles Gideon Korrell believes that the opinion serves as a reminder that physical realities of system design (such as the impossibility of true parallel beams) can be decisive in proving inherent anticipation. The court’s analysis—rooted in the inevitability of divergence and magnification in Jorgensen’s geometry—shows that anticipation can rest not only on what’s disclosed, but also on what must unavoidably occur.
By Charles Gideon Korrell