In In re Kostic, the Federal Circuit recently reinforced the statutory bar against broadening reissue applications filed beyond the two-year limit, emphasizing that claims must be construed based on their actual language rather than an inventor’s subjective intent.
Background:
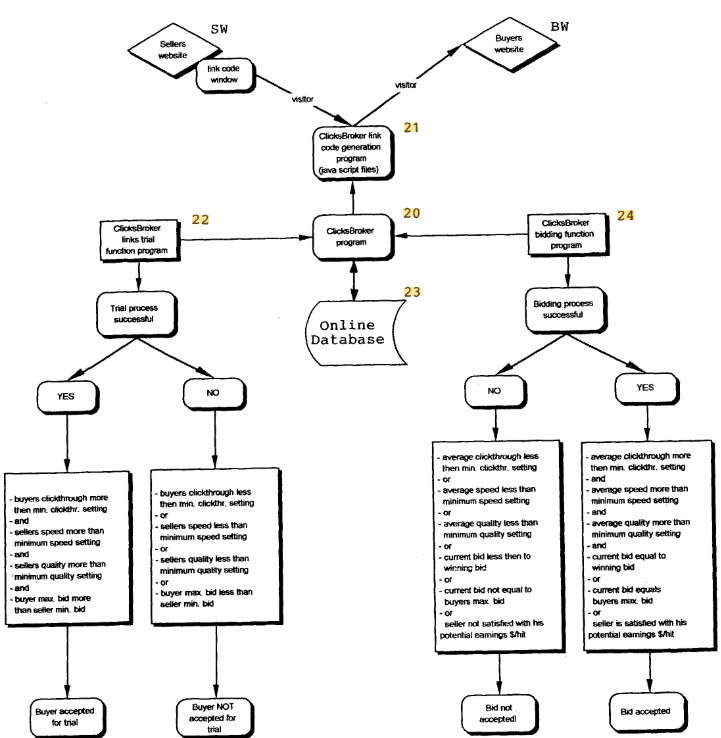
Appellants Kostic and Vandevelde sought reissue of U.S. Patent No. 8,494,950, covering methods for buying and selling click-through internet traffic via an intermediary website. Original dependent claim 3 allowed a direct traffic exchange without a trial process but explicitly depended on independent claim 1, which required a trial process. Arguing that original claim 3 was invalid under 35 U.S.C. § 112 for inconsistency, appellants attempted to rewrite it in independent form, providing optional pathways either with or without a trial process.
Federal Circuit Decision:
The Federal Circuit affirmed the PTAB’s rejection, holding the reissue claim was impermissibly broader than the original. The Court applied the principle established in Medtronic, Inc. v. Guidant Corp. that any claim containing at least one conceivable process not infringing the original claims is broader in scope.
Key Points of Law:
- Claim Construction: The Court clarified that the determination of claim scope for broadening reissues under 35 U.S.C. § 251(d) depends strictly on the claims as originally written, rather than on the patentees’ intended scope or subjective understanding. Citing Chef America, Inc. v. Lamb-Weston, Inc. and Superior Fireplace Co. v. Majestic Prods. Co., the Court underscored the importance of objective claim interpretation.
- Broadened Scope Analysis: Reissue claim 3 introduced optional pathways (with or without a trial process), whereas original claims mandated a trial process. Thus, the reissue claim was broader, capturing methods that would not infringe original claims. Since this broadened claim was sought beyond the two-year statutory limit, it violated 35 U.S.C. § 251(d).
Implications:
This decision reiterates the importance of careful drafting and review during patent prosecution. Inventors and practitioners must ensure that dependent claims clearly align with independent claims and avoid ambiguous constructions. Once the two-year period for broadening reissues passes, any attempts to retroactively broaden claims are statutorily barred, emphasizing the critical nature of claim precision at the outset.
Conclusion:
In re Kostic serves as a reminder of the rigorous statutory limitations surrounding reissue applications. Patent holders must rely on the explicit language of their claims rather than intentions or interpretations developed post-issuance. Clarity in original claim drafting remains paramount.
By Charles Gideon Korrell